
Electrophotocatalytic Generation of Unstabilized Ketyl Radicals and their Coupling to Alkenes
Contributors: Benjamin List, Luc M. Debie
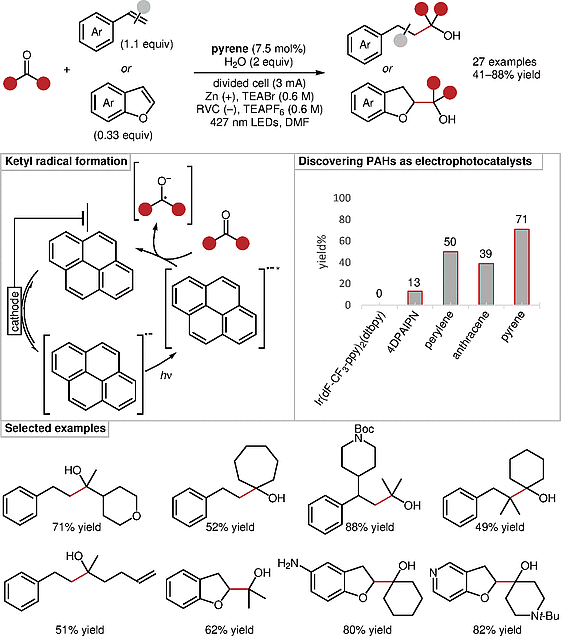
Significance
Wickens and co-workers report an electrophotocatalytic method of selectively generating unstabilized ketyl radicals, and their coupling to styrenes and benzofurans. The resulting coupling products were obtained in generally good yields. Control experiments in the absence of light showed that radical coupling is favored over hydrogenation only under the combined action of light and current, suggesting parallel reaction pathways.
Comment
The authors’ report expands upon previous applications of polycyclic aromatic hydrocarbons (PAHs) as photo- and electrocatalysts by combining these functionalities into one protocol. PAHs show superior reactivity to more established iridium-based and organic photocatalysts, partially due to PAHs’ increased stability. This stability is evidenced by the complete consumption of 4DPAIPN after just 13% product formation, as opposed to the recovery of 75% of the pyrene catalyst after full conversion.
Edgecomb JM, Alektiar SN, Cowper NG. W, Sowin JA, Wickens ZK. * University of Wisconsin−Madison, USA
Ketyl Radical Coupling Enabled by Polycyclic Aromatic Hydrocarbon Electrophotocatalysts
J. Am. Chem. Soc. 2023; 145: 20169-20175
DOI: 10.1021/jacs.3c06347.
